Photo AI
In an experiment to measure the heat of reaction (\Delta H) for the reaction between hydrochloric acid (HCl) and sodium hydroxide (NaOH), 100 cm³ of 2M HCl were added to 100 cm³ of 2M NaOH in the apparatus shown in the diagram - Leaving Cert Chemistry - Question 3 - 2012
Question 3
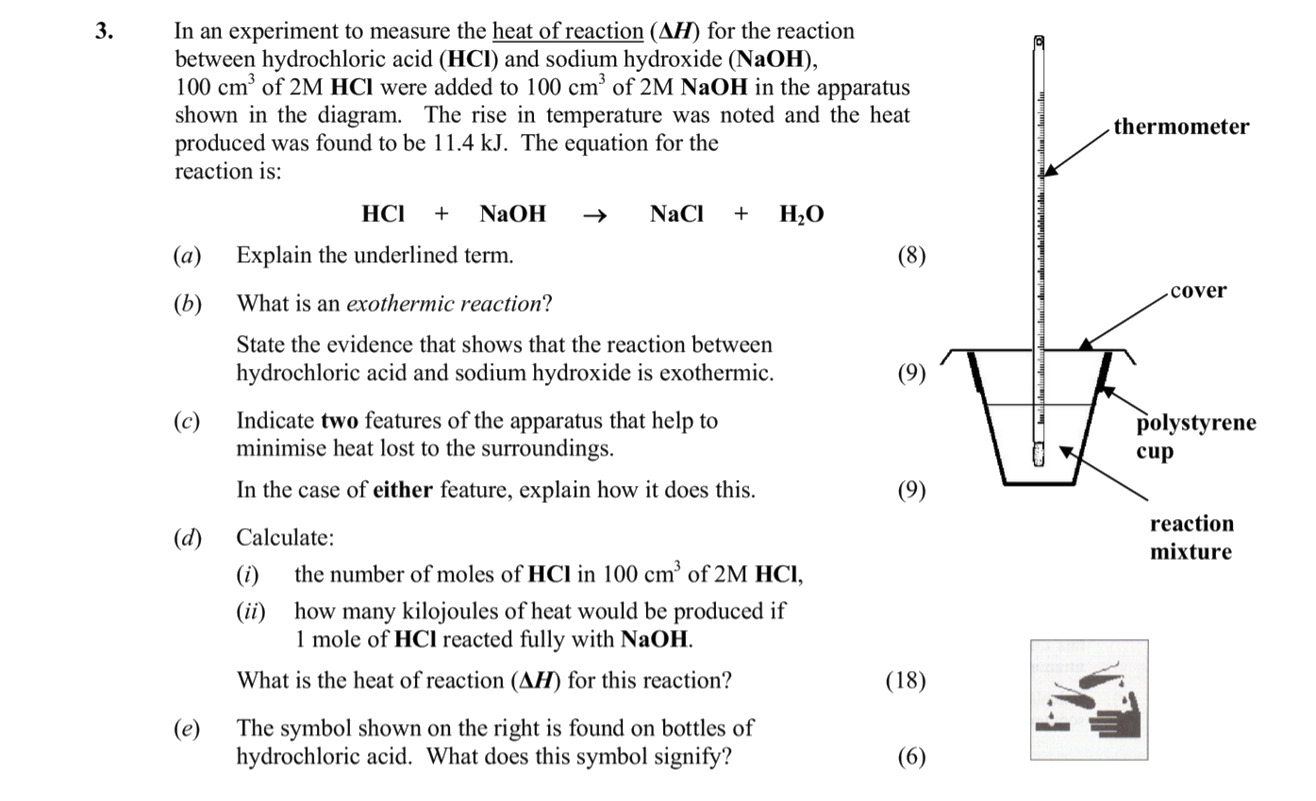
In an experiment to measure the heat of reaction (\Delta H) for the reaction between hydrochloric acid (HCl) and sodium hydroxide (NaOH), 100 cm³ of 2M HCl were adde... show full transcript
Worked Solution & Example Answer:In an experiment to measure the heat of reaction (\Delta H) for the reaction between hydrochloric acid (HCl) and sodium hydroxide (NaOH), 100 cm³ of 2M HCl were added to 100 cm³ of 2M NaOH in the apparatus shown in the diagram - Leaving Cert Chemistry - Question 3 - 2012
Step 1
Explain the underlining term.
Answer
The term refers to the heat change when one mole of HCl reacts completely with one mole of NaOH in the reaction. This indicates that the reaction is a neutralization process where these reactants produce salt (NaCl) and water (H₂O), resulting in a temperature change.
Step 2
Step 3
Indicate two features of the apparatus that help to minimise heat loss to the surroundings.
Answer
- Polystyrene Cup: The polystyrene cup is a poor conductor of heat, which helps to reduce heat loss.
- Cover: The cover on the cup provides insulation by preventing heat from escaping upward, thus retaining more heat within the system.
Step 4
Calculate the number of moles of HCl in 100 cm³ of 2M HCl.
Answer
The calculation for the number of moles is as follows:
Number of moles = concentration (mol/dm³) × volume (dm³)
Converting 100 cm³ to dm³ gives:
100 cm³ = 0.1 dm³
Therefore,:
Number of moles of HCl = 2 mol/dm³ × 0.1 dm³ = 0.2 moles.
Step 5
How many kilojoules of heat would be produced if 1 mole of HCl reacted fully with NaOH?
Answer
To find the heat produced for 1 mole of HCl:
- Heat produced from 0.2 moles of reaction: 1.14 kJ
- Heat per mole = \frac{1.14 ext{ kJ}}{0.2 ext{ moles}} = 5.7 ext{ kJ/mol}
Therefore, for 1 mole of HCl, it produces 5.7 kJ of heat.
Step 6
Step 7
